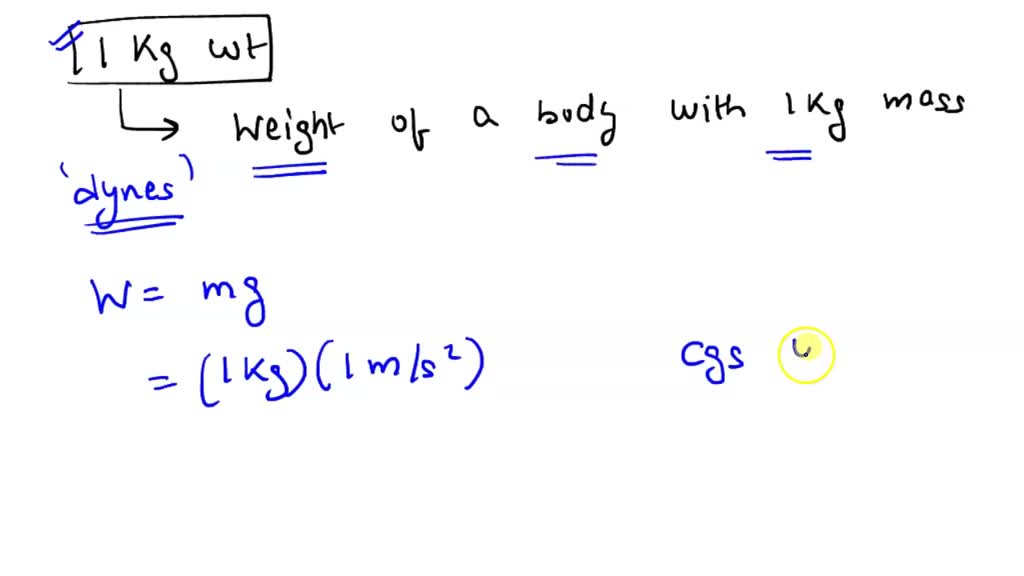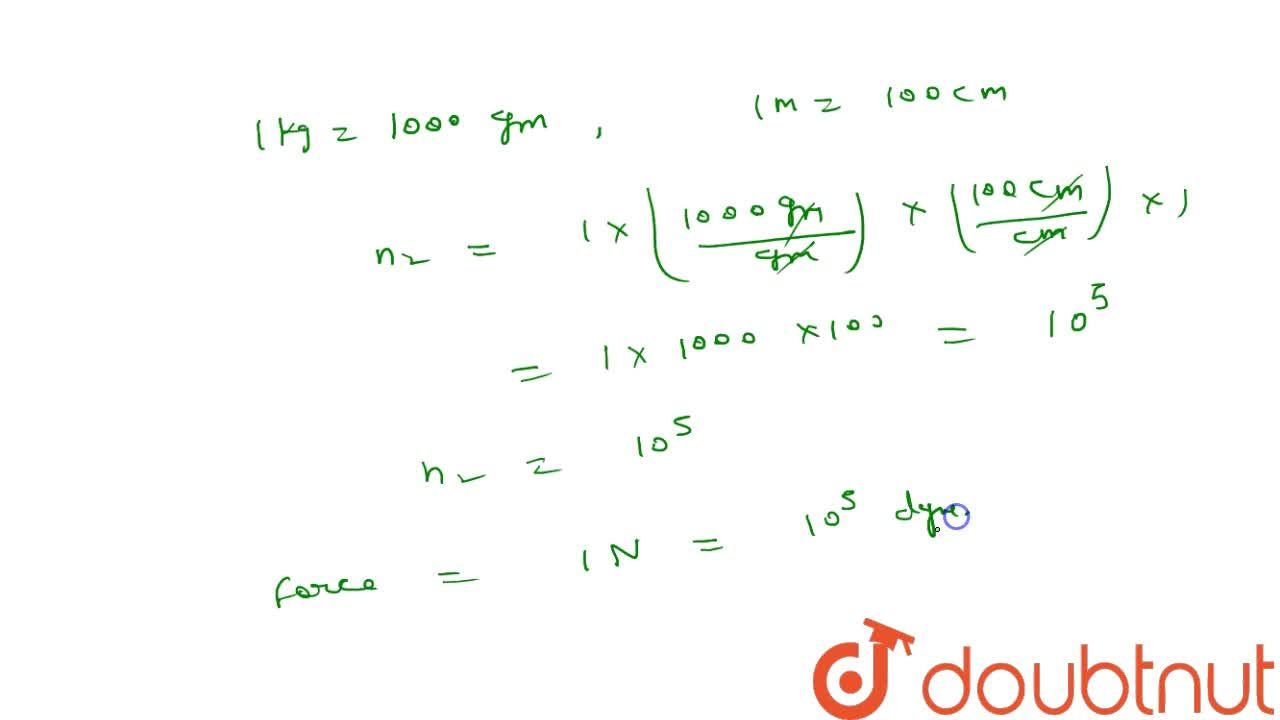If at a pressure of 10^6 dyne/cm^2 , one gram mole of nitrogen occupies 2 × 10^4 c.c. volume, then the average energy of nitrogen molecules in erg is :

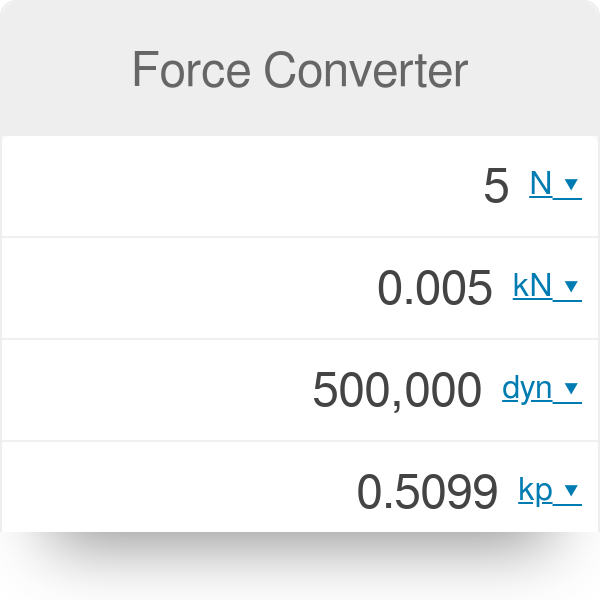
The SI units of force is newton such that 1N="1kg ms"^(-2). In C.G.S. system, force is expressed in dyne. How many dyne of force is equivalent to a force of 5 N?

Activity 1: solve for size and speed of a drop.. Useful units for calculations Remember: dyne = 1 g cm/s 2 (force in cm-g-s units) = Newtons. Using. - ppt download